How to choose a CRO
Why choosing a CRO can make or break a project
Current Status of Outsourcing in Non-clinical and Clinical Trials
In recent years, the development of new drugs, such as biopharmaceuticals, gene therapy, and regenerative medicineHighly specialized due to diversification of modalitiesThe company's own assets (facilities and personnel) are no longer sufficient to complete all testing. It is no longer possible to complete all tests using only one's own assets (facilities and personnel), and reliance on external specialized organizations with specific skills and know-how has never been greater. Modern CROs are not simply "outsourcers" of work,An important "strategic partner" to share scientific knowledge and accelerate development togetherThe role of the company is changing to
Three risks of CRO selection errors (increased costs, delayed delivery, and lower quality)
Improper CRO selection can be fatal to a project.
First,Redoing the exam due to inexperience with the techniquewill increase costs by tens of millions of yen.
Second, due to inadequate progress managementdelivery delaydelays the submission to the PMDA/FDA and directly leads to the loss of market exclusivity opportunities, which are estimated at several hundred million yen per day.
Third,Poor quality in terms of unreliability of datacan lead to the worst possible outcome: rejection of the application for approval or suspension of development.
Choosing solely on the basis of "short-term cheapness" is an extremely high-risk gamble.
5 criteria for selecting a "no-fail" CRO
1. expertise and experience
Strong in oncology," "proven track record in the central nervous system," etc,In-depth knowledge of specific disease areas and study typesMake sure that you have the following information. In addition to the number of past contracts, academic support through publications and academic activities is also important. If they have extensive experience in conducting similar study protocols, they are a trustworthy partner with the ability to avoid unexpected problems.
(2) Facilities and equipment
State-of-the-art 3D image analysis and highly sensitive mass spectrometer,Is the necessary equipment available for the test?are essential items to check. On the other hand, more importance should be placed on strict animal husbandry management, a backup system in case of power outages, and the degree of support for digital data management (e.g., CSV support) rather than the age of the facility. On sites where equipment is thoroughly maintained, data accuracy will naturally dwell as well.
3. reliability and quality assurance
Strict adherence to GLP standardsas well as an independent quality assurance (QA) department to ensure that it is functioning properly. especially when international submissions to the U.S. FDA and European EMA are being considered,Experience with inspections by foreign authoritiesThe success or failure of a project depends on the company's transparency and transparency. A highly transparent approach to information disclosure, such as the status of improvements to past findings, is also a major factor in the selection process.
4. communication and flexibility
The scientific background of the person in charge and the speed of response to problems directly affect the quality of the test. Do not just passively wait for instructions,Do you have professional suggestions for optimizing the study design?Determine what is the best way to achieve the project's goals. A relationship in which the contractor does not "throw the project away," but rather engages in scientific discussions with each other and allows for flexible adjustments, will lead to the success of the project.
5. cost-effectiveness
It is dangerous to make a decision based solely on the low initial estimate.Conditions for incurring additional costsPlease compare by "total cost," which includes the cost of the study and the certainty of a quick and successful study. The "second opinion cost" of requesting a low-cost CRO, not being able to use the data, and having to re-test the data at another company is a significant loss.Final Return on Investment (ROI)It is necessary to have an eye for calmly discerning the
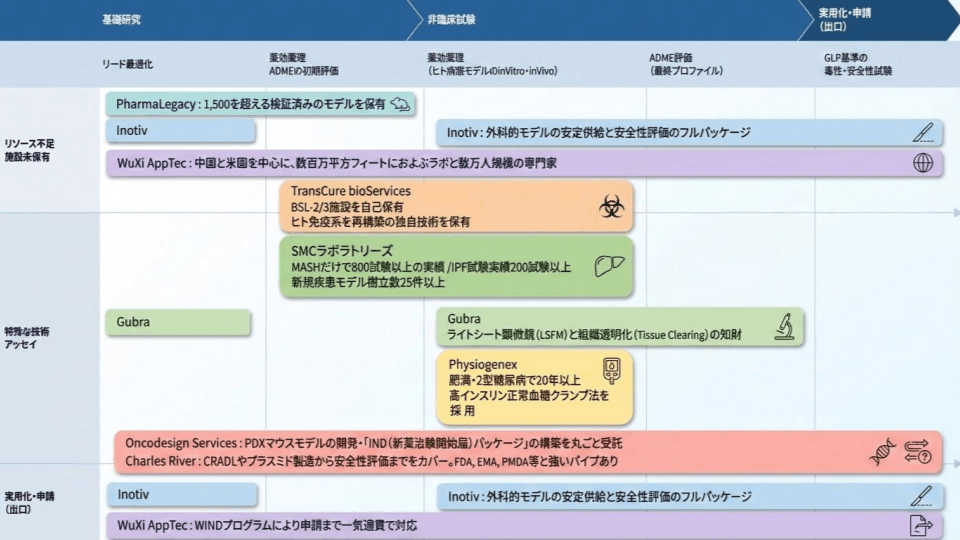
Positioning map and feature comparison of major CROs
The development phase of a drug discovery study begins with basic research, followed by initial evaluation, drug efficacy and pharmacology studies in ADME and human disease models, and finally GLP-standard toxicity and safety studies before moving to FIT. The CRO to be selected depends on the phase of drug discovery research and the company's own issues.
We have selected nine companies with standard models that cannot be dismissed in the current drug discovery trend (metabolism and oncology) and independent companies that are globally recognized for their specific advanced technologies.
Survey period: January-March 2026.
Comparison of Major CRO Features
| Disease area |
SMC Laboratories, Inc. |
Physiogenex SAS |
Gubra A/S |
Oncodesign Services |
TransCure BioServices |
PharmaLegacy |
WuXi AppTec |
Inotiv, Inc. |
Charles River Laboratories, Inc. |
| feature |
Test Performance: Over 800 tests with MASH alone
IPF Test Results: 200+ Tests
Establishment of new disease models: 25+ cases
|
Obesity and Type 2 diabetes for over 20 years
Adopt the euglycemic hyperinsulinemic clamp technique |
Intellectual Property of Light Sheet Fluorescence Microscopy (LSFM) and Tissue Clearing |
Outsourced development of PDX mouse models and complete construction of the IND (Investigational New Drug) application package |
Self-owned BSL-2/3 facility
Possesses proprietary technology for rebuilding the human immune system. |
Over 1,500 verified models |
Millions of square feet of labs and tens of thousands of specialists, primarily in China and the United States
Handles the entire process from application to completion with the WIND program. |
A full package for stable supply and safety evaluation of surgical models. |
Covers everything from CRADL and plasmid manufacturing to safety assessment. Strong relationships with FDA, EMA, PMDA, etc. |
| Inflammation |
O |
- |
- |
O |
O |
O |
O |
O |
- |
| Fibrosis |
O |
O |
O |
- |
- |
O |
O |
O |
- |
| Liver disease (MASH/HCC) |
O |
O |
O |
- |
O |
O |
O |
O |
- |
| Tumors (including IO) |
O |
- |
- |
O |
O |
O |
O |
O |
O |
| Respirator |
- |
- |
O |
- |
O |
O |
O |
O |
O |
| Central Nervous System |
O |
- |
O |
O |
- |
O |
O |
O |
O |
| Metabolism |
O |
O |
O (Obese) |
- |
Mars |
O |
O |
O |
- |
| autoimmunity |
O |
O |
- |
Rheumatism |
O |
O |
O |
O |
O |
| Official HP |
https://www.smccro-lab.com/jp/ |
https://www.physiogenex.com/ |
https://www.gubra.dk/ |
https://www.oncodesign-services.com/ |
https://www.transcurebioservices.com/ |
https://www.pharmalegacy.com/ |
https://www.wuxiapptec.com/ |
https://www.inotiv.com/ |
https://www.criver.com/ |
Difference between major domestic, independent, and foreign companies and how to use them
Major foreign companies have strength in simultaneous global applicationsbut tend to be costly and small. On the other handMajor domestic companies are PMDA compliantThe company is highly trusted by the government of Japan and is familiar with the essentials of domestic applications. In additionIndependents specialize in specific assaysand can respond quickly and flexibly. The optimal use of the system must be determined by the size and purpose of the project.
Advantages of using a boutique CRO that is strong in a specific area
Exclusive ownership of specific disease models (e.g., STAM mice for NASH/MASH)CROs that specialize in the early phases of drug discovery and small "boutique" CROs with specialized technologies have unique strengths. In the early phases of drug discovery, which require "sharp technology" that cannot be imitated by large companies, these specialized organizations provide unparalleled testing efficiency and highly accurate data. The greatest advantage is that they can derive solutions to niche issues in the shortest possible time.
Specific steps from CRO selection to the start of outsourcing
Step 1: Create RFP (Request for Proposal) and define requirements
The first step in selecting a CRO is to prepare a Request for Proposal (RFP) that clearly states the purpose, duration, budget, and required data accuracy for the study. ThisIf the definition of requirements is unclear, the assumptions used in the quotations submitted by the various companies will vary, making it impossible to make appropriate comparisons.Providing detailed information on the level of deliverables your company requires is the key to eliciting highly accurate proposals.
Step 2: Sign Non-Disclosure Agreement (NDA) and narrow down candidates
Promptly sign a Non-Disclosure Agreement (NDA) prior to disclosing specific information. Then, conduct initial hearings with several companies based on the RFP.Confirm expertise, estimated costs, and ability to meet deadlinesBy properly screening out companies that do not meet your company's requirements at this stage, you can greatly increase the efficiency of the next step, the facility tour and in-depth evaluation.
Step 3: Points to check during the facility tour (audit)
During the facility tour, the cleanliness of the site, calibration labels on equipment, raw data storage, and staff training records are rigorously checked.Particularly important is the nonverbal information about whether the people on the ground are proud of their work.It is. In addition to compliance with procedures (SOPs), attention to detail and integrity are essential factors that determine the reliability of data.
Step 4: Comparison of quotations and final decision
In the final stage, each company'sComprehensive evaluation of the appropriateness of the proposal (test design and methods) as well as the priceThe following is a brief overview of the process. Instead of simply choosing the cheapest, examine whether the proposed METHOD is scientifically optimal and whether the risk measures are adequate. It is important to select, on the basis of evidence, the one company whose balance of price and quality best matches your company's project strategy.
Key Points in Selecting a CRO for Global Expansion
Availability of overseas offices and strength of network
We will check to see if local testing overseas is possible and if the local partner network is strong. In particular,Logistics system for international transportation of biological samples and experience in handling customs clearance problemsis an important matter that affects the global examination schedule.
Multilingual support and knowledge of foreign regulatory authorities
As well as the ability to write quality reports in English,Experience in supporting responses to inquiries (queries) from FDA and EMAFocus on the following. A CRO that is well versed in the requirements of foreign authorities will give you a strong edge in entering the global marketplace.
In summary: Find the best partner for your company's needs.
Success or failure in selecting a CRO is an extremely important process that determines the speed and quality of new drug development. Please sort out your company's project size, therapeutic areas, and future global expansion plans, and then compare them to the five selection criteria introduced in this article to determine the most appropriate partner.